|
Mixtures have a variable mass ratio depending upon what quantities of ingredients have been combined in the mixture. This is true of all pyrite no matter the sample size. pyrite has 46.6% iron and 53.4% sulphur by mass. Mixtures can be homogeneous or non-homogeneous.Ĭompounds have specific mass ratios. Solids, Liquids and Gases can be combined to create a mixture. Compounds are classified into molecular compounds, ionic compounds, intermetallic compounds and complexes. chocolate milk retains properties of chocolate and milkĪ huge, virtually limitless, number of chemical compounds can be created. Mixture: two or more different atoms together but not joined. Compounds are chemical substances that are made up of two or more atoms of the same or different elements that are linked together by chemical linkages. They reflect the properties of their constituent substances, which retain their original properties. Compound: two or more different atoms bonded together. Mixtures do not have specific, consistent chemical and physical properties of their own. 
Mixtures cannot be represented by chemical formulas.Ĭompounds have specific chemical and physical properties that are distinct from their constituent elements because the constituent elements lose their properties when they combine to make the compound. Salt in water pasta and sauce sand pebbles solutions such as rubbing alcohol, dental amalgam, vapor in air colloids such as mayonnaise, milk, cheese coarse suspensions such as mud in water, oil in water.Ī compound is represented using its chemical formula that represents the symbols of its constituent elements and the number of atoms of each element in one molecule of the compound. Water (H2O), Sodium chloride (NaCl), Sodium bicarbonate (NaHCO3) and Hydrochloric acid (HCl) are examples of compounds. Often they are visibly distinct.Ī compound can be separated into simpler substances by chemical methods/reactions.Ī mixture can be separated into simpler substances by physical or mechanical methods. Under certain conditions, water also forms a supercritical fluid. 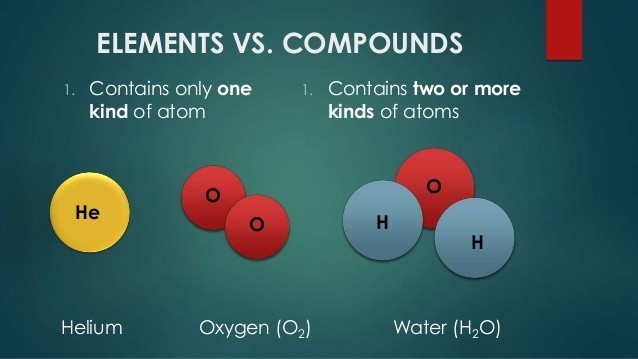 
The solid phase is known as ice and the gas phase is called steam. The name water typically refers to the liquid state of the compound. The ingredients are physically mixed but chemically separate. Water is a chemical compound consisting of two hydrogen atoms and one oxygen atom. Mixtures contain different elements and compounds but the ratio is not fixed nor are they combined via chemical bonds. Elements that compose the compound are chemically combined. In a compound, elements are chemically bonded together, which makes it very difficult to separate them. Comparison chart Compound versus Mixture comparison chartĪ compound contains atoms of different elements chemically combined together in a fixed ratio.Ī mixture is a combination of two or more substances where there is no chemical combination or reaction.Ĭompounds contain different elements in a fixed ratio arranged in a defined manner through chemical bonds. A compound is a pure substance that is made from more than one element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
